:max_bytes(150000):strip_icc()/carbonatom-58b602855f9b5860464c8bf6.jpg) 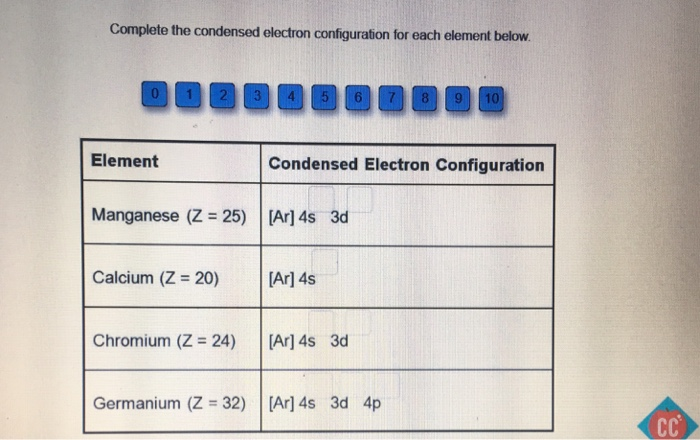
For example, 1s2 2s2 should be entered as 1s 22s 2. Express your answer in complete form in order of increasing orbital energy. Therefore the Li electron configuration will be 1s 2 2s 1. Since 1s can only hold two electrons the remaining electron for Li goes in the 2s orbital. In writing the electron configuration for lithium the first two electrons will go in the 1s orbital. Use the rules for determining electron configurations to write the electron configuration for Ca. Lithium is the third element with a total of 3 electrons. and 2p are the occupied subshells, and the superscript "2" is the number of electrons in each of these subshells. 6.7: Many-Electron Atoms 6. The electron configuration of the carbon atom represented by the orbital diagram is This electron configuration can be written as 1s2 2s2 2p2 where 1s, 2s. The total number of electrons in a neutral carbon atom is 6. For example, consider a carbon atom having an atomic number of 6. From the electrons in an atom, to the differing orbitals and hybridization, the ground state electron configuration sheds light on many different atomic properties. The electron configuration can also be represented by writing the symbol for the occupied subshell and adding a superscript to indicate the number of electrons in that subshell. Ground state electron configurations are the foundation for understanding molecular bonding, properties, and structures. For example, 4s23d8 would be entered as 4s 23d 8. Express your answer in condensed form as a series of orbitals. Give the ground-state electron configuration for copper (Cu) using noble-gas shorthand. Give the condensed electron configuration for Ti4+ (Z 22). Express your answer in condensed form as a series of orbitals. Give the condensed electron configuration for La3+ (Z 57). We fill both the 1 s and 2 s orbitals to achieve a 1 s2 2 s2 electron configuration: When we reach boron, with Z 5 and five electrons, we must place the fifth electron in one of the 2 p orbitals. The next element is beryllium, with Z 4 and four electrons. In writing the electron configuration for carbon the first two electrons will go in the 1s orbital. This electron configuration is written as 1 s2 2 s1. Give the ground-state electron configuration for silicon (Si) using noble-gas shorthand. Carbon is the sixth element with a total of 6 electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
